Post‑Exposure Prophylaxis: Mitoquinol as a Novel Defense Against COVID‑19
Written by Georgia Truman (MSc), Molecular and Cellular Biology. Reviewed by Dr. Siobhan Mitchell (PhD), Neuroscience.
Post‑exposure prophylaxis options for SARS‑CoV‑2 remain limited, particularly for individuals whose immune responses to vaccination are impaired. While most approaches focus on suppressing viral replication once infection is established, far fewer strategies address the host‑cell processes that shape early immune control. Rather than targeting the virus directly, this exploratory study examined whether preserving mitochondrial immune signalling could alter infection outcomes following confirmed high‑risk exposure.
What you’ll learn
How mitochondrial function underpins early innate antiviral immune responses
Why SARS‑CoV‑2 disrupts mitochondrial signalling to evade immune detection
The impact of Mitoquinol on infection rates and symptom duration after high‑risk exposure
Implications for prophylaxis in immunocompromised and unvaccinated populations
What role do mitochondria play in immune signalling?
To understand the basis of this approach, it is first necessary to consider the role of mitochondria in antiviral defence. Beyond their metabolic functions, mitochondria act as central signalling hubs in innate immunity. Mitochondrial antiviral signalling protein (MAVS), anchored to the outer mitochondrial membrane, coordinates interferon production following viral RNA detection.
This process depends on intact mitochondrial membrane potential and redox balance. When mitochondrial function is compromised, the speed and amplitude of antiviral signalling decline, widening the window for viral replication. As a result, mitochondrial health influences infection susceptibility and disease severity, rather than merely reflecting downstream immune activation.
What factors influence the virulence of SARS‑CoV‑2?
SARS‑CoV‑2 has evolved mechanisms that directly exploit this vulnerability. During early infection, viral proteins disrupt mitochondrial membrane potential, fragment the mitochondrial network, and inhibit MAVS‑dependent interferon signalling. In parallel, elevated mitochondrial oxidative stress further impairs immune detection while amplifying dysregulated inflammatory responses.
These effects are particularly consequential in immunocompromised and unvaccinated individuals, where immune redundancy is reduced and early signalling failures carry greater clinical cost. This makes preservation of mitochondrial function especially relevant in populations with limited capacity to compensate for early innate immune disruption.
How might Mitoquinol reduce SARS‑CoV‑2 infection rate and severity?
Against this backdrop, Mitoquinol was evaluated for its potential to stabilise mitochondrial signalling during the earliest stages of viral exposure. Mitoquinol accumulates within the mitochondrial matrix via its triphenylphosphonium targeting group, where it scavenges superoxide and preserves mitochondrial membrane stability.
By reducing oxidative disruption of mitochondrial dynamics, Mitoquinol supports intact MAVS signalling and promotes a more rapid interferon response. Importantly, this mechanism does not inhibit viral replication directly, but instead alters the intracellular environment that enables immune evasion during early exposure — a critical distinction for post‑exposure prophylaxis.
How effective was Mitoquinol in preventing SARS‑CoV‑2 infection?
To determine whether these cellular effects translated into clinical benefit, investigators conducted a pragmatic open‑label study involving 80 adults with confirmed high‑risk SARS‑CoV‑2 exposure. Participants initiated supplementation shortly after exposure, allowing the intervention to intersect with the early phase of viral replication.
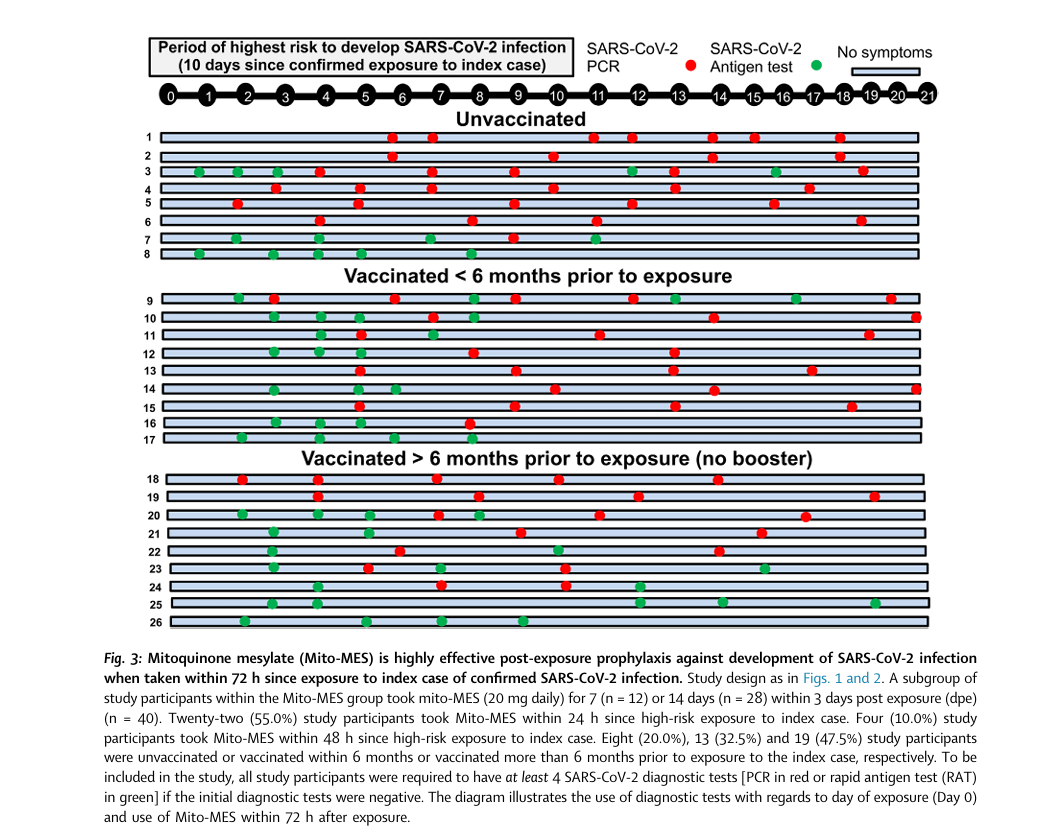
Within this real‑world setting, 30% of participants receiving Mitoquinol (20 mg/day) developed SARS‑CoV‑2 infection, compared with 75% of matched controls (odds ratio 0.17).
Among participants who did become infected, median symptom duration was shorter in the Mitoquinol group (3 days versus 5 days), indicating benefit even when infection prevention was incomplete. Together, these findings suggest that mitochondrial support may influence both infection establishment and clinical course.
Image taken from Chen et al., 2024.
What are the implications for high‑risk populations?
Importantly, the study cohort included unvaccinated cancer patients and individuals living with HIV — groups for whom vaccine‑derived protection may be attenuated and prophylactic options remain limited. Within these subpopulations, Mitoquinol demonstrated consistent reductions in infection rates and symptom burden.
Given its oral availability and established safety profile, these findings support further evaluation of mitochondria‑targeted approaches as adjunctive prophylaxis in settings where immune protection is incomplete or compromised.
What should practitioners know about dosing and timing?
Intervention began within 72 hours of exposure, aligning with the period during which mitochondrial immune signalling exerts the strongest influence on infection outcomes. The 20 mg daily dose mirrors established safe use in cardiovascular and metabolic trials, supporting its feasibility for short‑term prophylactic use.
Beyond acute exposure, the results raise broader questions regarding sustained prophylaxis and potential applications in mild COVID‑19 or post‑acute sequelae, where persistent mitochondrial dysfunction has been increasingly implicated.
Read the full article:Mitoquinoluinone mesylate as post-exposure prophylaxis against SARS‑CoV‑2 infection in humans – PubMed DOI: 10.1016/j.ebiom.2024.105042