What is Mitoquinol Mesylate?
Mitoquinol mesylate (commercially known as MitoQ®) is a mitochondria‑targeted antioxidant developed to selectively reduce oxidative stress within mitochondria—the primary intracellular source of reactive oxygen species (ROS).
Originally developed by Professor Mike Murphy and Dr Robin Smith, Mitoquinol was designed to overcome the poor mitochondrial uptake of conventional antioxidants and to directly test the role of mitochondrial oxidative stress in aging and disease. Today, mitoquinol is one of the most extensively studied mitochondrial research compounds, with broad application across preclinical and human clinical research.
How it works
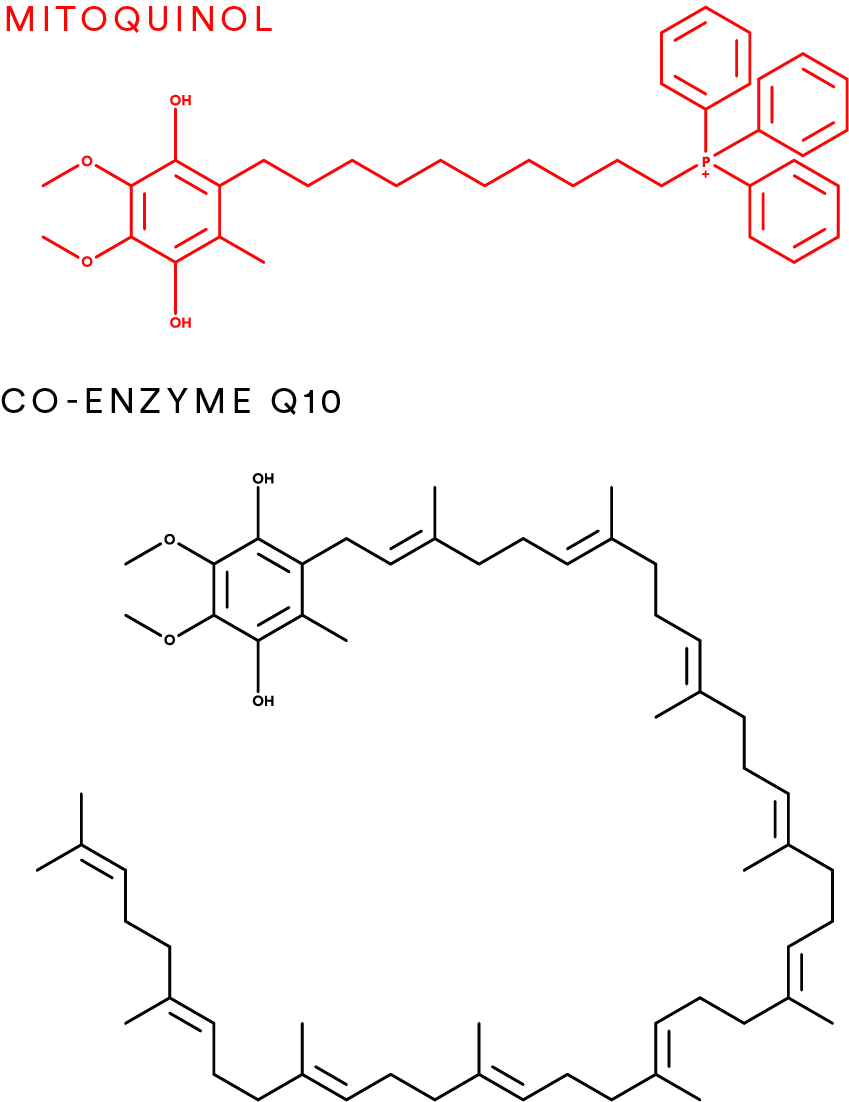
Mitoquinol is a modified form of coenzyme Q10 (CoQ10) designed to overcome the poor mitochondrial uptake of conventional CoQ10 supplements.
Structurally, mitoquinol consists of:
A ubiquinol antioxidant headgroup (the reduced, active form of CoQ10)
A lipophilic triphenylphosphonium (TPP⁺) cation, linked via a ten‑carbon alkyl chain
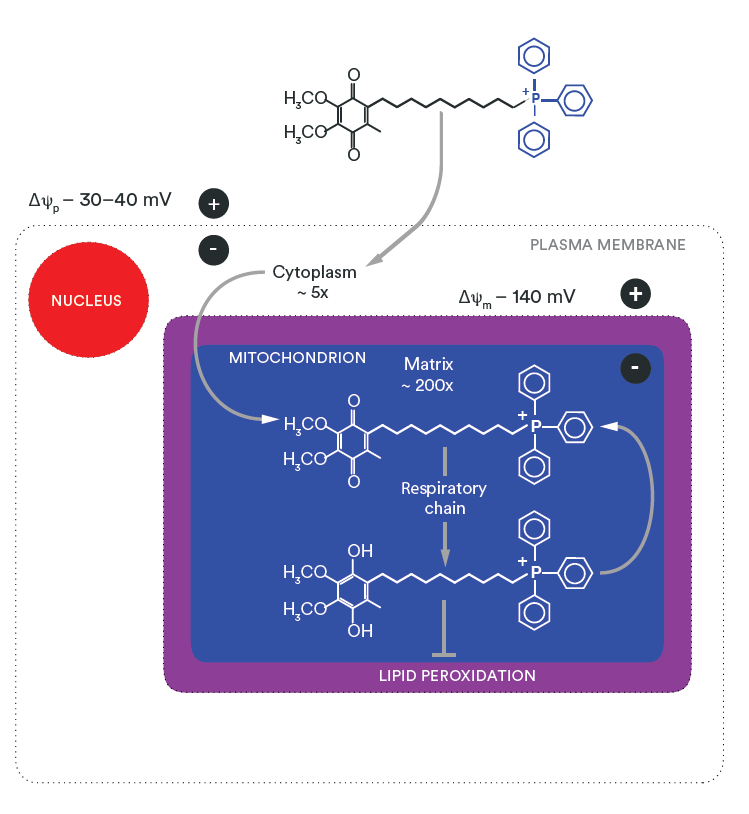
This design allows mitoquinol to exploit the strong negative electrical potential of the inner mitochondrial membrane, driving selective and rapid mitochondrial accumulation after oral or experimental administration.
Once inside mitochondria, mitoquinol localises to the inner mitochondrial membrane, positioning it directly at the site of electron transport chain activity and mtROS generation.
Mechanism of Action
How mitoquinol targets mitochondria
Mitoquinol’s mechanism of action is defined by three interrelated properties:
Selective mitochondrial accumulation
The positively charged TPP⁺ moiety drives mitoquinol into mitochondria, where it accumulates hundreds‑fold relative to cytosolic concentrations. Uptake is especially high in metabolically active tissues such as heart, skeletal muscle, brain, liver, and kidneys
Direct and indirect antioxidant activity
Within the inner mitochondrial membrane, mitoquinol:
Neutralises mitochondrial ROS (including superoxide and peroxynitrite)
Reduces lipid peroxidation of mitochondrial membranes
Protects mitochondrial and nuclear DNA from oxidative damage
In addition, mitoquinol has been shown to increase endogenous antioxidant capacity, up‑regulating enzymes such as catalase, glutathione peroxidase, and superoxide dismutase.
3. Redox recycling and sustained activity
After neutralising ROS, mitoquinol is oxidised to mitoquinone but is rapidly recycled back to its active form by mitochondrial complex II. This recycling allows sustained antioxidant activity at relatively low doses, distinguishing mitoquinol from non‑recyclable antioxidants.
Clinical Trial Evidence: Human Research Summary
Mitoquinol has been evaluated in multiple randomised, placebo‑controlled human clinical trials, most commonly at daily doses of 10–20 mg. Across studies, mitoquinol demonstrates good tolerability and consistent biological effects aligned with mitochondrial redox modulation.
Cardiovascular and vascular aging research
Clinical studies in older adults and cardiometabolic populations report improvements in:
Endothelial function and flow‑mediated dilation
Oxidized LDL cholesterol
Aortic stiffness and vascular oxidative stress markers
These findings support the role of mitochondrial oxidative stress in vascular aging and endothelial dysfunction.
Exercise and mitochondrial bioenergetics
Human trials show mitoquinol reduces:
Exercise‑induced mitochondrial DNA damage
Post‑exercise oxidative stress biomarkers
Importantly, mitoquinol does not blunt physiological exercise adaptations, making it valuable for studying redox signalling rather than indiscriminate antioxidant effects.
Inflammation and oxidative stress biomarkers
Across clinical datasets, mitoquinol is associated with reductions in:
Lipid peroxidation markers (e.g. F2‑isoprostanes, MDA)
Inflammatory mediators such as IL‑6
Increases in total antioxidant capacity
Preclinical Research Applications
Mitochondrial dysfunction and aging biology
Preclinical studies show mitoquinol influences:
Mitophagy and mitochondrial quality control
AMPK, mTOR, sirtuin, and PGC‑1α signalling pathways
Mitochondrial respiration and oxidative resilience
These mechanisms underpin its use in aging and longevity research models.
Reproductive and developmental biology
Mitoquinol has been studied in fertility and developmental models, where it:
Protects oocytes and sperm from oxidative damage
Improves mitochondrial function during in vitro maturation
Enhances developmental competence under hypoxic or metabolic stress
Cardiometabolic and inflammatory disease models
Cellular and animal studies show mitoquinol attenuates:
Endothelial dysfunction
Cardiac hypertrophy and fibrosis
Mitochondrial damage in metabolic stress contexts
Why Use Mitoquinol in Mitochondrial Research?
For researchers and clinicians, mitoquinol offers:
A mechanistically precise mitochondrial intervention
Extensive preclinical and human validation
Predictable pharmacokinetics and bioavailability
Compatibility with biomarker‑driven study designs
Direct relevance to human physiology and aging
Crucially, mitoquinol is best positioned not as a generic antioxidant, but as a mitochondria‑specific experimental tool for interrogating redox biology in translational research.
Mitoquinol and the MCRP
The MitoQ Clinical Research Program (MCRP) supports investigator‑led research into mitochondrial health and disease. Researchers may explore opportunities for:
Compound and placebo access
Trial design support
Biomarker integration
Collaborative mitochondrial research