Does Mitoquinol Protect DNA from Damage?
Written by Georgia Truman (MSc), Molecular and Cellular Biology. Reviewed by Dr. Siobhan Mitchell (PhD), Neuroscience.
High‑intensity exercise induces a transient surge in mitochondrial reactive oxygen species. While moderate oxidative signalling is required for adaptation, excessive mtROS can damage nuclear and mitochondrial DNA. This study assessed whether chronic Mitoquinol supplementation could mitigate DNA damage without blunting exercise‑induced adaptation.
What you'll learn:
How high‑intensity exercise induces mitochondrial and nuclear DNA damage
Why indiscriminate antioxidant use can impair training adaptation
The differential effects of acute versus chronic Mitoquinol supplementation
How Mitoquinol influences DNA damage markers without suppressing adaptive signalling
How does DNA damage occur during intense exercise?
During high‑intensity exercise, elevated energy demand increases electron flux through the mitochondrial respiratory chain. As workload rises, so does the probability of electron leak, resulting in increased production of superoxide. When this oxidative load exceeds the cell’s buffering capacity, reactive species interact with nucleic acids, producing oxidative lesions in DNA.
Mitochondrial DNA is particularly vulnerable in this context. Its close proximity to the inner mitochondrial membrane, combined with the absence of protective histones and more limited repair capacity, makes it especially susceptible to oxidative damage. Accumulated lesions can impair mitochondrial function over time, undermining both energy production and long‑term cellular adaptation to training.
What role does mitochondrial signalling play in exercise adaptation?
Importantly, not all exercise‑induced oxidative stress is harmful. Transient increases in mtROS act as signalling molecules that activate key adaptive pathways, including PGC‑1α‑mediated mitochondrial biogenesis and Nrf2‑regulated antioxidant defence. These responses underpin improvements in endurance, metabolic efficiency, and resilience to repeated stress.
Problems arise when oxidative stress is suppressed indiscriminately. Studies using conventional antioxidants such as vitamins C and E have shown that blunting ROS broadly can impair these adaptive responses. An effective intervention must therefore reduce pathological oxidative excess while leaving physiological signalling bursts intact.
How does Mitoquinol prevent exercise‑induced DNA damage?
Mitoquinol was evaluated in this context because of its unique mitochondrial targeting properties. By accumulating selectively within the mitochondrial matrix, Mitoquinol scavenges excess superoxide at its primary source. Crucially, this localisation allows mitigation of DNA‑damaging mtROS without interfering with cytosolic or nuclear redox signalling pathways that drive adaptation.
In theory, this positions Mitoquinol to strike a balance between protection and plasticity — reducing oxidative injury while preserving the signalling environment required for training‑induced benefit.
What was the impact of Mitoquinol on DNA damage during intense exercise?
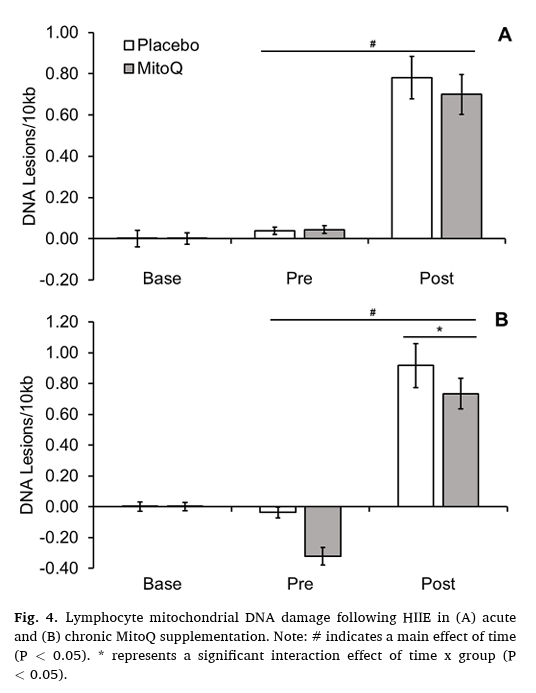
To test this hypothesis, investigators examined DNA damage responses in 24 healthy men undergoing high‑intensity exercise. Participants received either acute supplementation or chronic Mitoquinol supplementation (20 mg/day for 21 days), allowing assessment of whether duration of exposure influenced mitochondrial protection.
Chronic supplementation significantly reduced exercise‑induced DNA lesions in both nuclear and mitochondrial DNA compared with placebo, whereas acute supplementation had no protective effect — consistent with the requirement for mitochondrial accumulation.
Quantitatively, DNA damage increased by approximately 15.9% in the chronic Mitoquinol group, compared with ~28.9% in the placebo group. These findings demonstrate meaningful genomic protection across both circulating lymphocytes and skeletal muscle tissue.
Image taken from Williamson et al., 2020.
Why was chronic supplementation more effective than a single dose?
This difference reflects the pharmacokinetic properties of Mitoquinol. Sustained dosing is required for the compound to accumulate within mitochondrial membranes and reach concentrations sufficient to buffer oxidative stress effectively. A single dose does not allow adequate mitochondrial loading and therefore cannot counter acute oxidative bursts during exercise.
The results indicate that Mitoquinol confers protection through pre‑emptive mitochondrial positioning rather than reactive scavenging, reinforcing the importance of supplementation strategy when targeting mitochondrial processes.
What are the practitioner takeaways for athletic health?
Notably, markers of adaptive signalling and cardiovascular response were unchanged, indicating that training‑relevant oxidative signalling was preserved. This reinforces the distinction between mitochondrial‑targeted protection and nonspecific antioxidant suppression.
When patients begin exercise‑based therapies—particularly those who are older, deconditioned, or managing chronic disease—there is often a transient increase in oxidative stress as mitochondrial activity and oxygen utilization rise before adaptive antioxidant defenses are fully upregulated. This early phase of exercise adaptation is biologically normal, but if oxidative stress is excessive or poorly buffered, it can contribute to muscle fatigue, delayed recovery, vascular strain, and reduced adherence to prescribed exercise programs.
Protecting against oxidative stress with mitoquinol mesylate during this initiation period supports healthier mitochondrial function, preserves DNA integrity, and helps maintain vascular and metabolic stability, allowing patients to gain the therapeutic benefits of exercise while reducing unnecessary physiological stress. As a result, oxidative stress protection is increasingly viewed as an important supportive strategy alongside exercise prescriptions for cardiovascular, metabolic, and age‑related conditions.
Read the full article:The mitochondria‑targeted antioxidant Mitoquinol attenuates exercise‑induced mitochondrial DNA damage – PubMed
DOI: 10.1016/j.redox.2020.101673