Safety First: Confirming the Absence of Nephrotoxicity at High Doses
Written by Georgia Truman (MSc), Molecular and Cellular Biology. Reviewed by Dr. Siobhan Mitchell (PhD), Neuroscience.
As mitochondria‑targeted antioxidants move into broader clinical investigation, organ safety at high doses becomes a critical consideration. In vitro studies had suggested potential renal toxicity at extreme concentrations, raising questions that required direct testing in humans.
What you’ll learn:
Why renal safety is a key concern for mitochondria‑targeted compounds
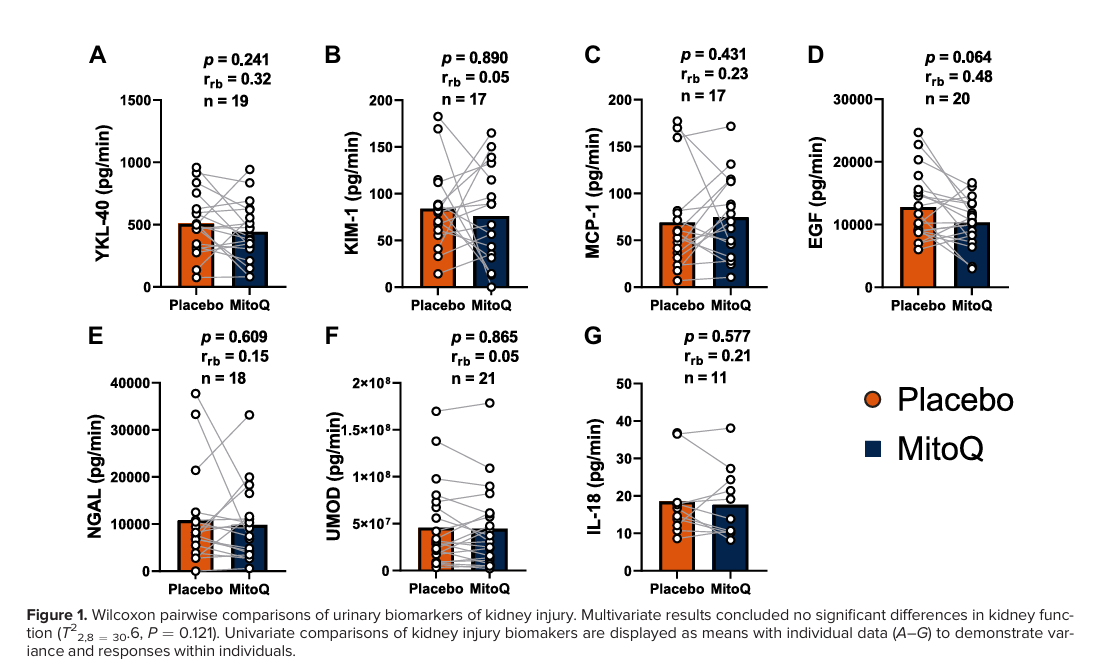
The effects of supratherapeutic Mitoquinol doses on kidney function biomarkers
What sensitive injury markers reveal beyond standard clinical measures
Why in vitro toxicity findings do not necessarily predict human risk
Why does nephrotoxicity testing matter for mitochondria‑targeted therapies?
The kidneys are among the most metabolically active organs in the body and are highly dependent on intact mitochondrial function to support filtration, reabsorption, and electrolyte regulation. Compounds engineered to accumulate within the mitochondrial matrix therefore warrant careful evaluation for renal safety, particularly at higher doses.
Concerns surrounding Mitoquinol originated from isolated renal cell culture studies, where exposure to very high static concentrations produced cytotoxic effects. However, such systems lack the clearance, metabolism, and tissue‑level regulatory mechanisms present in whole organisms. This made it essential to assess renal effects under physiologically relevant conditions in humans.
What does high‑dose Mitoquinol do to kidney function?
To address this question, investigators conducted a randomised crossover study in which 32 healthy adults received acute, weight‑based Mitoquinol doses ranging from 100 to 160 mg — up to eight times higher than commonly studied therapeutic doses.
Renal function was assessed using standard clinical measures, including creatinine clearance, urine flow rate, and electrolyte balance. Across all parameters, no significant differences were observed between Mitoquinol and placebo, indicating preserved kidney function even under supratherapeutic exposure.
What did specialised kidney injury markers reveal?
Recognising that conventional clinical markers can miss subtle or early renal injury, the study incorporated a panel of sensitive biomarkers to detect subclinical nephrotoxicity. These included kidney injury molecule‑1 (KIM‑1), neutrophil gelatinase‑associated lipocalin (NGAL), and YKL‑40, analysed individually and using multivariate statistical approaches.
No significant changes were detected in any biomarker, either alone or in combination. This absence of signal indicates that even at doses deliberately chosen to stress safety margins, Mitoquinol did not induce detectable nephron‑level injury.
Image taken from Linder et al., 2023.
Why don’t in vitro toxicity findings always translate to humans?
In vitro systems expose cells to static compound concentrations without the compensatory mechanisms present in vivo. There is no hepatic metabolism, renal clearance, tissue distribution, or adaptive response to mitigate intracellular accumulation.
In humans, by contrast, Mitoquinol is filtered and excreted through the kidneys, preventing the sustained intracellular exposure seen in cell culture models. This trial underscores why human safety studies are indispensable for interpreting theoretical risks suggested by preclinical systems.
What is the practitioner takeaway for safety?
Taken together, these findings provide robust evidence that Mitoquinol does not induce kidney injury across the entire dose range used in clinical research — and well beyond it.
For clinicians and researchers, this establishes a substantial safety margin at commonly studied doses (20–40 mg/day) and places earlier in vitro concerns into appropriate context. As mitochondrial‑targeted therapies continue to expand into new indications, this study provides essential reassurance that renal safety is preserved even under conservative worst‑case exposure conditions.
Read the full article:Acute high‑dose Mitoquinol does not increase urinary kidney injury markers in healthy adults – American Journal of PhysiologyDOI: 10.1152/ajprenal.00186.2023